Clinical Research Process
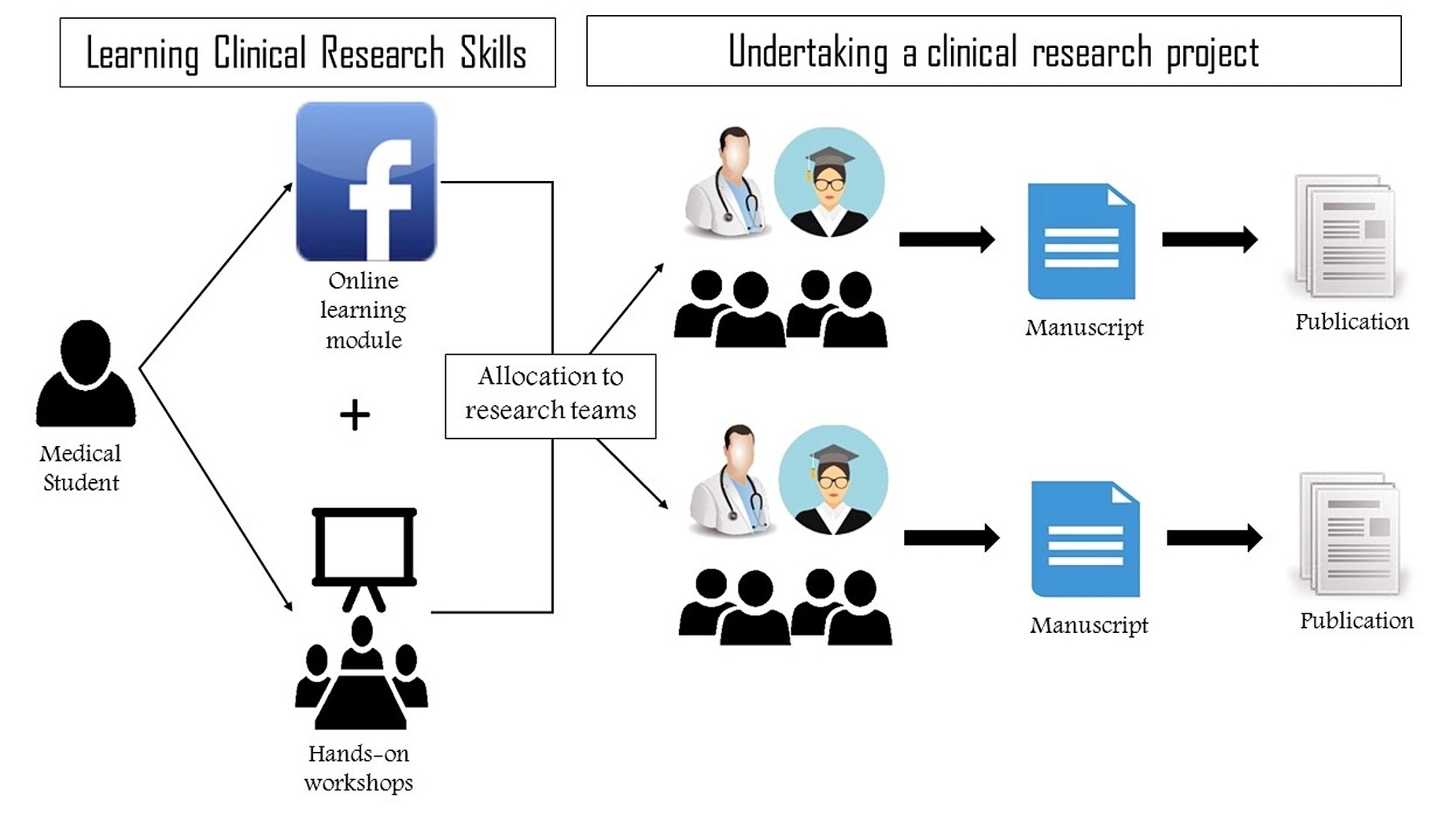
Clinical research refers to studies, or trials, that are done in people. As the developers design the clinical study, they will consider what they want to accomplish for each of the different Clinical Research Phases and begin the Investigational New View Diagram Clinical Research Process
